What Certifications Do You Need to Import and Sell Cosmetics in the US? (FDA, GMP, and Labeling)
-
 Yulia Blinova
Yulia Blinova
- Updated: May 27, 2026
- 18 min read
Many new cosmetics importers assume the United States is a light-touch market compared to the EU. That is no longer accurate. Since the Modernization of Cosmetics Regulation Act of 2022 (MoCRA) became enforceable on July 1, 2024, every brand selling skincare, makeup, hair products, or other cosmetics in the US has to deal with mandatory FDA facility registration, product listing, evolving GMP expectations, safety substantiation, adverse event reporting, and strict labeling rules under MoCRA.
This guide breaks down what compliance actually looks like. You will learn what the FDA expects from your facility, your product, and your label, where the most expensive mistakes happen, and how to build compliance into your sourcing strategy from day one rather than fixing problems at customs later.
Why cosmetics compliance in the US matters more than people think
Compliance is not about where you produce cosmetics. It is about where you sell it. A serum filled in Korea, a lipstick made in Italy, or a cream produced in China all face the same FDA framework once they enter the United States.
The right mental model is risk management for your money, your inventory, and your brand. When a shipment is held at the port, the cost is not just storage fees. It is delayed launches, missed marketing windows, retailer penalties, and frozen working capital sitting in a container.
Common business consequences when cosmetics compliance is incomplete include customs holds (shipments blocked at the US port), Amazon and retailer delistings (marketplaces require valid registration and labeling), forced recalls under MoCRA, civil and competitor risk including facility suspension, and loss of buyer trust with distributors and retailers who often require proof of FDA registration before they will accept a pitch.
Brands that build compliance into the sourcing brief from day one move faster, get into more retail channels, and avoid the expensive cycle of relabeling and re-shipping. The ones that bolt it on at the end almost always pay more, both in fees and in lost time.

What counts as a cosmetic in the US?
Before you can know which certifications you need, you have to know what category your product falls into. The FDA defines cosmetics as articles intended to be applied to the human body for cleansing, beautifying, promoting attractiveness, or altering appearance. Practically, that includes skincare (cleansers, moisturizers, serums, masks), color cosmetics (foundation, lipstick, mascara, eyeliner), hair products (shampoo, conditioner, styling products), fragrance, and personal care items like deodorants and bath products.
The trap: some products look like cosmetics but are legally regulated as drugs. Anti-acne treatments, sunscreens, anti-dandruff shampoos, antiperspirants, and any product making a treatment claim (“treats wrinkles,” “cures acne,” “blocks UV”) will be classified as either a drug or a cosmetic-drug combination. That triggers an entirely different set of requirements, including OTC monograph compliance and stricter manufacturing oversight. If your label leans into medical claims, your compliance pathway changes dramatically.
The 5 pillars of US cosmetics compliance under MoCRA
Cosmetics compliance in the US now rests on five pillars. Miss one and your product is not legally marketable, regardless of how good the formulation is.

1. FDA facility registration
Every facility that manufactures or processes cosmetics for sale in the US must be registered with the FDA. This applies to both domestic and foreign facilities. Foreign factories must designate a US agent who can receive FDA communications and assist with inspections. Registration requires obtaining an FDA Establishment Identifier (FEI) before submission, and registrations renew every two years, which functions as the facility’s unique identifier in all FDA records.
If you are sourcing private label or contract manufacturing in Asia or Europe, this is the first concrete supplier question to ask: “Are you MoCRA registered, and can you share your FEI number?” If they cannot answer, that is a red flag.
2. Product listing
Each individual cosmetic product sold in the US must be listed with the FDA. The responsible person, meaning the manufacturer, packer, or distributor whose name appears on the label, must list each marketed cosmetic product, including ingredients, and update annually. Listings are submitted electronically through Cosmetics Direct using SPL (Structured Product Labeling) format.
For importers, the key point is this: someone has to be the responsible person. If your name is on the label, that is usually you. The legal weight for safety, listing accuracy, and adverse event reporting sits with the responsible person, not with the foreign factory.
3. Good Manufacturing Practices (GMP)
GMP is about how the product is made, not just what is in it. While the FDA is still finalizing MoCRA-specific GMP regulations, the international benchmark is ISO 22716, which already covers personnel training, facility hygiene, raw material control, production records, batch traceability, packaging integrity, complaint handling, and internal audits.
In practical terms, your factory must be able to show documented standard operating procedures, batch records that trace from raw material to finished unit, stability and microbial testing on file, calibrated equipment, and trained staff in proper hygiene zoning. If a supplier cannot show this paperwork, walk away.
4. Safety substantiation
Every cosmetic sold in the US now requires the responsible person to maintain records that substantiate the safety of the product. This is one of the most overlooked parts of MoCRA. It does not mean “the ingredients are generally safe.” It means you have documented evidence that this specific formula, in this specific packaging, used as directed, is safe.
Typical safety substantiation files include toxicological assessment of each ingredient, stability testing (typically 3 months accelerated, 12 months real-time), preservative challenge testing, microbial testing on finished product, heavy metals testing for products with mineral pigments, and patch testing or HRIPT (Human Repeat Insult Patch Test) where relevant.
5. Adverse event reporting
The responsible person must report serious adverse events to the FDA within 15 business days and keep records for six years (three for small businesses) and maintain records of all adverse events (serious and non-serious) for six years (three for small businesses). A serious adverse event is anything resulting in death, life-threatening reaction, hospitalization, persistent disfigurement, or requiring medical intervention. As an importer, you need a documented intake process from day one, even if your sales volume is small.

Cosmetics labeling requirements for the US market
Even with FDA registration, GMP, and product listing in place, your product can still be blocked or delisted if the label is wrong. US cosmetics labels are governed by the FD&C Act and the Fair Packaging and Labeling Act (FPLA), and they have very specific rules.
A compliant US cosmetics label must include:
- Product identity (what the product is)
- Net quantity in both US units (oz, fl oz) and metric (g, mL)
- Name and place of business of the responsible person
- Ingredient declaration in descending order using INCI names
- Country of origin (“Made in China,” “Made in Italy,” etc.)
- Required warnings or directions (especially for eye-area products)
A few traps that catch importers:
- Fragrance allergens: new disclosure rules for specific allergens are expected through 2026.
- Talc-containing products: standardized asbestos testing rules are being finalized.
- Drug claims hidden in marketing copy: “anti-aging cream” might pass; “removes wrinkles” can be reclassified as a drug.
One of the most expensive avoidable mistakes is discovering a non-compliant label after production has been completed. Re-stickering a container of cosmetics is a logistical nightmare and often costs more than the original print run.

Country strategy: where you source affects compliance
Compliance does not just live in paperwork. It lives in the maturity of the factory you choose. Different sourcing regions trade off cost, GMP maturity, and documentation readiness in different ways. The right choice depends on your category, volume, brand positioning, and exit plans.
| Region | Cost | GMP Maturity | Documentation Readiness | Best For |
|---|---|---|---|---|
| China | Lowest | Varies widely. Top-tier factories can match EU standards. | Requires strict supplier verification and contract structure. | High-volume, cost-sensitive sourcing with strong oversight. |
| Vietnam / Southeast Asia | Competitive | Mid-range, improving. Many factories export-ready. | Generally good for export. | Reducing China’s tariff exposure. Zignify case: $300K–$500K annual savings vs. China. |
| Korea | Higher than China | Strong, especially for skincare. | Generally good. | Premium skincare and K-beauty formulations. |
| Italy, France, Germany, Poland | Highest | Highest globally. Traceability and compliance are built in. | Full traceability, ingredient compliance standard. | Premium retail brands and exit-focused businesses. Zignify case: €1.4M savings in 8 months on €25M production. |
China
Most cost-efficient by a clear margin, but GMP maturity varies dramatically between factories. Top-tier Chinese cosmetics manufacturers can match European standards. Mid-tier factories often need significant work to meet MoCRA expectations. Strict supplier verification, on-site sample inspection, and a clear contract structure are non-negotiable.
Vietnam and Southeast Asia
Increasingly used by US importers wanting to reduce China exposure due to tariffs. Cost is competitive, GMP maturity is mid-range and improving, and many factories are export-ready. In a Zignify case, moving cosmetics-related production from China to Vietnam delivered $300,000 to $500,000 in annual savings while improving supplier structure.
Korea
Strong cosmetics ecosystem, especially for skincare and K-beauty formulations. Documentation is generally good. Cost is higher than in China, but quality and innovation often justify it for premium brands.
Italy, France, Germany, Poland
Highest GMP maturity globally. Documentation, traceability, and ingredient compliance are baked in. Cost is significantly higher, but for brands targeting premium retail or planning an exit, this can dramatically increase brand value. In one Zignify cosmetics project at €25M production with an exit in mind, sourcing was distributed across China, Germany, France, Italy, and Poland (glass bottles, jars, tins, pumps, packaging from Miron-Glas France, a German production company, bottles and pumps from Italy), and packaging optimization plus better suppliers delivered €1.4M in savings within 8 months.

Step-by-step: how a US cosmetics import actually works
This is the realistic sequence we walk clients through when they want to launch a cosmetics line into the US market. Skipping a step rarely saves money. It usually costs more later.
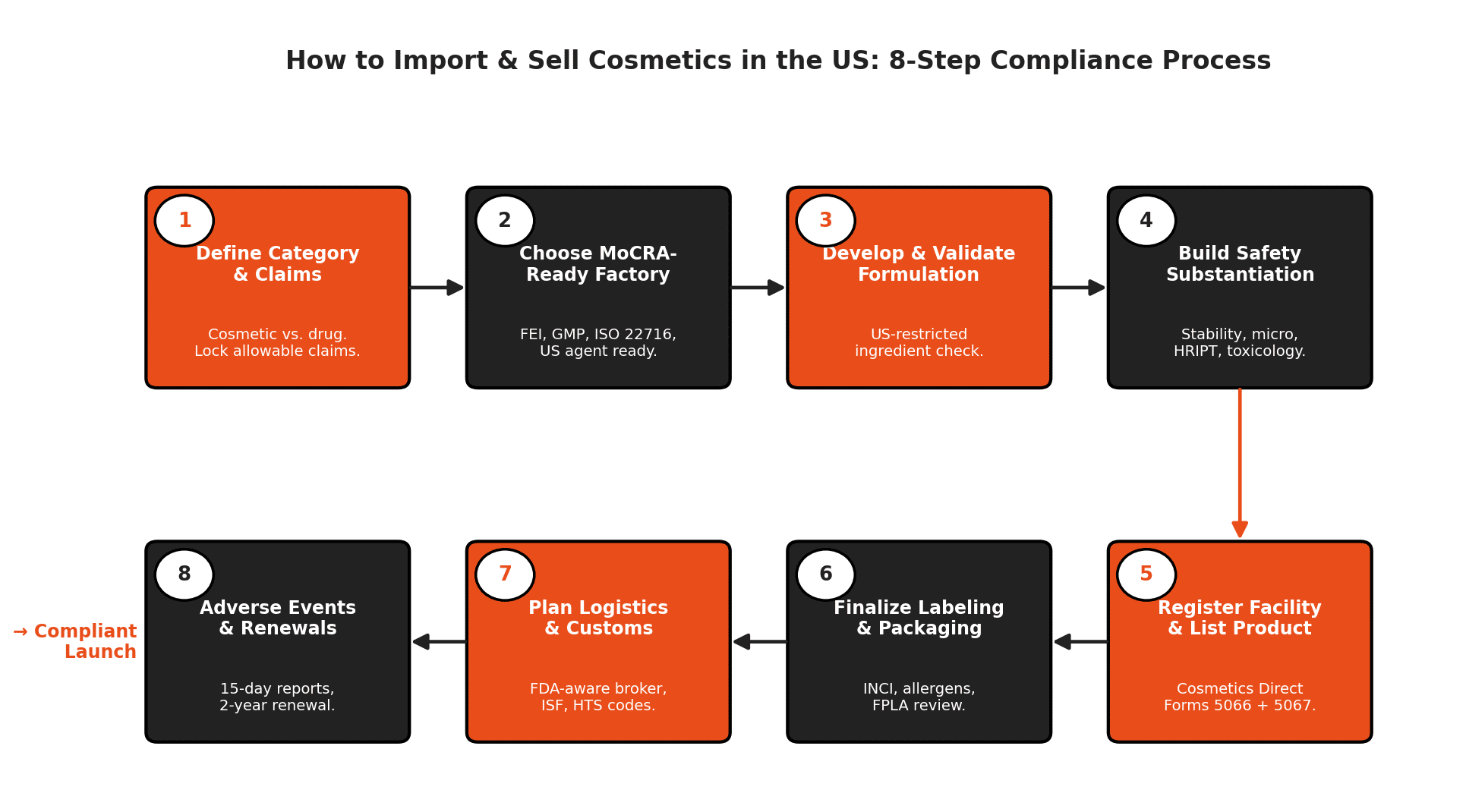
Step 1: Define product category and claims
Lock in whether your product is a cosmetic, an OTC drug, or a combination. Decide what claims you can legally make. This decision drives every other compliance step.
Step 2: Choose a manufacturing partner with compliance in mind
Not every contract manufacturer is MoCRA-ready. Ask for FDA facility registration confirmation and FEI number, ISO 22716 certificate or equivalent GMP documentation, stability and challenge test reports, and US agent details if the factory is foreign. A factory that has never exported to the US before is not automatically disqualified, but you will need to budget more time and money.
Step 3: Develop and validate the formulation
Work with a formulator who understands US restricted and prohibited ingredients. Some ingredients allowed in the EU or Asia are restricted or prohibited in the US, and vice versa. FDA testing of imported gel nail polish removers in 2024 and 2025 detected methylene chloride contamination, which usually traces back to inadequate raw material control.
Step 4: Build the safety substantiation file
Commission stability testing, preservative challenge testing, microbial testing, heavy metals testing where relevant, patch testing or HRIPT for skin contact products, and a toxicological risk assessment by a qualified toxicologist. Keep these records for the full FDA retention period.
Step 5: Register the facility and list the product
Once the factory has its FEI, the responsible person submits facility registration (Form FDA 5066) and product listing (Form FDA 5067) through Cosmetics Direct. This should be completed before the first shipment leaves the origin country to avoid delays or compliance issues at entry. A product entering the US without a valid listing is non-compliant under MoCRA and exposed to enforcement risk.

Step 6: Finalize labeling and packaging
Lay out the label in compliance with FD&C Act and FPLA requirements. Have it reviewed by someone who has done US cosmetics labeling before. Run the artwork past your supplier with the exact ingredient list, allergen declarations, country of origin, and net quantity statement. Lock the artwork before printing.
Step 7: Plan logistics, customs, and import documentation
US Customs and Border Protection (CBP) coordinates with the FDA on cosmetics imports. Your importer of record, broker, and ISF filing all need to align with the registration data. Build your shipping plan with a freight forwarder who has handled FDA-regulated cosmetics, not just generic consumer goods.
Step 8: Set up adverse event tracking and ongoing compliance
Once products are on the market, log every customer complaint and assess for adverse event reporting. Renew your facility registration every two years. Update your product listing whenever ingredients change. Monitor upcoming MoCRA rules on fragrance allergen labeling, asbestos testing, and any future ingredient bans.

Common cosmetics import mistakes that cost money
Across hundreds of sourcing conversations, the same expensive mistakes keep showing up.
Mistake 1: Assuming supplier compliance equals your compliance
A foreign factory might be MoCRA-registered, but they are not your responsible person. The FDA looks at whose name is on the label. If something goes wrong, your business takes the hit, not theirs.
Mistake 2: Choosing suppliers without regulatory experience
This leads directly to wrong labeling, missing documentation, and customs issues. A factory making cosmetics for the local Chinese market may have no concept of INCI naming or US allergen disclosure. They will quote a great price and produce a non-compliant product.
Mistake 3: Trusting only the first sample
Cosmetics scale up from lab to mass production in ways that change viscosity, color, scent, and microbial behavior. Brands that approve a sample, place 50,000 units, and skip pre-shipment inspection regularly receive product that looks slightly off. Define a documented golden sample with measurable parameters, then inspect production batches against it before final payment moves.
Mistake 4: Ignoring packaging compliance
Packaging is part of regulation, not just branding. A cream stable in glass might oxidize in plastic. A serum fine in clear bottles might degrade in UV. Heavy metal migration from low-grade packaging is a real issue. Packaging optimization has also delivered some of the biggest savings in Zignify cosmetics projects, including double-digit raw material cost reductions.
Mistake 5: No verification of certificates
Many suppliers claim certifications they do not actually hold, or hold expired ones. “GMP certified” can mean anything from a real audited certificate to a printed PDF with no traceable issuer. Always verify.
Five mistakes drain millions from cosmetics importers every year. They are predictable. We help you avoid them by building each step into the sourcing plan from day one. Book a free sourcing call →

How Zignify supports cosmetics importers
Zignify is not a manufacturer, a trading company, or a label printer. The role is closer to an experienced operator sitting on your side of the table while you build a compliant cosmetics supply chain. There are no factory commissions or kickbacks, and pricing is transparent (typically hourly, sometimes share-of-savings).
For cosmetics importers entering the US, our support typically covers:
- Supplier search and verification: scanning broad pools of cosmetics manufacturers across China, Vietnam, Korea, Italy, France, Germany, and Poland. Filtering for MoCRA-ready facilities with the right GMP track record.
- Compliance research: mapping every certification, test, and label requirement specific to your product category, then turning that into a clear roadmap before production.
- Documentation and certificate verification: confirming GMP certificates, ingredient compliance, and safety documentation are real and current.
- Lab testing management: identifying suitable testing labs, comparing quotes, coordinating sample submission, and making sure test reports are issued in your company’s name.
- Sample and production quality control: inspecting samples in country before they reach you, defining a golden sample, and inspecting production batches before final payment.
- Purchase agreement support: shaping contracts that cover penalties for delays, who pays for QC, payment terms, and what happens if compliance documents are missing.
- Logistics optimization: comparing freight forwarders rather than relying on the factory’s preferred partner often surfaces meaningful savings.
Zignify Real Case Study: Cosmetics Outcomes
- €1.4M in 8 months: a cosmetics brand at €25M production preparing for exit. Zignify sourced better suppliers across China, Germany, France, Italy, and Poland, optimized packaging (glass bottles, jars, tins, pumps from Miron-Glas France, a German production partner, raw material cost reduction for filler, bottles and pumps from Italy), and improved supplier quality. Result: €1.4M savings, ≈5.8% of revenue, profit jump from €2.5M to €3.9M, scaling within 3 years to exit.
- $300K to $500K annual savings: a US-market beauty accessories supply chain hit by tariffs and supplier issues. Zignify moved production to Vietnam, improved supplier structure, eliminated 27% import tariffs, and gained better delivery times and stronger margin going into exit.

What Most Guides Get Wrong, Here’s What Our Expert Knows
Most cosmetics import guides treat compliance as a checklist of forms. The reality is that most expensive failures happen between the steps, in the handoffs between the formulator, factory, label designer, and freight forwarder. Three patterns show up in almost every problem case.
A factory’s MoCRA registration does not transfer regulatory responsibility away from you
Importers see “MoCRA-registered factory” and assume the regulatory weight sits with the factory. It does not. The responsible person, the entity named on the label, carries safety substantiation, adverse event reporting, and listing accuracy. If a brand has the factory’s FEI but no internal safety file, no complaint log, and no qualified US agent contact, the brand gets the FDA letter, not the factory. Build your own compliance file from day one and treat the factory’s registration as a prerequisite, not a substitute.
The first production batch almost never matches the approved sample exactly
Cosmetics scale up from lab batches to thousands of units in ways that change viscosity, color stability, scent profile, and microbial behavior. Brands that approve a sample, place a 50,000-unit order, and skip pre-shipment inspection regularly receive product that looks slightly off, sometimes badly off. Define a documented golden sample with measurable parameters, then inspect the production batch against it before final payment moves. Anything else is paying first and hoping later.
Label print runs are the single most common source of rework cost
Printing tens of thousands of incorrect labels and then relabeling at port or in a US warehouse is one of the most expensive avoidable mistakes in cosmetics imports. Triggers are usually small: an ingredient declared in non-INCI form, a missing allergen, an incorrect net quantity, or a marketing claim that crosses into drug territory. Have the final label artwork reviewed against FDA and FPLA requirements before the print order is placed.
If you are about to commit to a cosmetics supplier or print run for the US market, it is worth pressure testing the plan before the money moves. Book a free sourcing call →
Frequently asked questions
1. Do I need FDA approval before I can sell cosmetics in the US?
The FDA does not approve most cosmetics before sale. What is required is FDA facility registration, product listing, GMP-aligned manufacturing, safety substantiation, and proper labeling. Color additives are an exception, and many require pre-market approval.
2. Who is the responsible person under MoCRA?
The responsible person is the manufacturer, packer, or distributor whose name appears on the cosmetic product label. For most foreign-made cosmetics sold in the US, this is the importing brand. They carry the legal weight for listing, safety, and adverse event reporting.
3. Does my foreign cosmetics factory need to register with the FDA?
Yes. Any facility, foreign or domestic, that manufactures or processes cosmetics distributed in the US must register with the FDA and renew every two years. Foreign facilities also need to designate a US agent.
4. What is GMP for cosmetics in the US?
GMP, or Good Manufacturing Practice, is the documented set of practices that govern how cosmetics are produced. The FDA is finalizing MoCRA-specific GMP regulations, but the global benchmark is ISO 22716, covering staff training, hygiene, raw material control, batch records, and quality assurance.
5. What does a compliant US cosmetics label require?
At minimum: product identity, net quantity in both US and metric units, name and place of business of the responsible person, ingredient declaration in INCI nomenclature in descending order, country of origin, and any required warnings or directions.
6. How to import cosmetics to the USA step by step?
Define product category and claims, choose a MoCRA-ready manufacturer, validate the formulation, build a safety substantiation file, register the facility and list the product through Cosmetics Direct, finalize compliant labeling, plan logistics, then set up ongoing adverse event tracking and renewals.
7. What happens if my product is found non-compliant after it is on the market?
Consequences range from warning letters and import alerts to forced recalls, facility suspension, and civil penalties. Marketplaces like Amazon also delist non-compliant products. Beyond regulatory action, brand and retailer trust take significant damage.
8. Are there exemptions for small cosmetics businesses under MoCRA?
Yes. Certain small businesses are exempt from facility registration and product listing. However, exemptions do not apply to facilities making products that come into regular contact with eye mucous membranes, are injected, or are intended to alter appearance for more than 24 hours. Labeling, safety substantiation, and adverse event reporting still apply broadly.
9. Can I sell cosmetics on Amazon in the US without FDA registration?
In practice, no. Amazon increasingly requires proof of FDA registration and compliance documentation for cosmetic listings. Even if a product slips through initially, an enforcement sweep or competitor complaint can take it down quickly.



